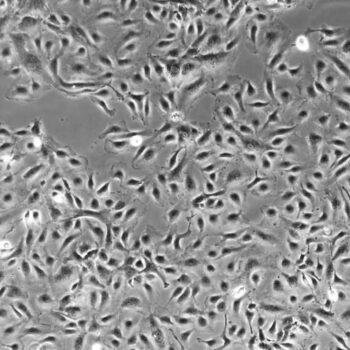
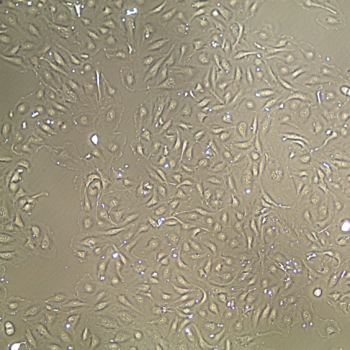
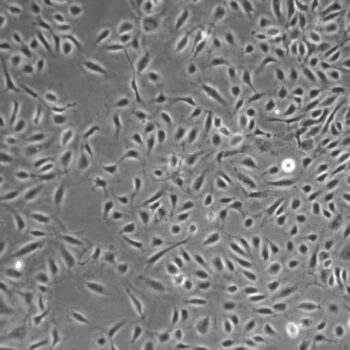
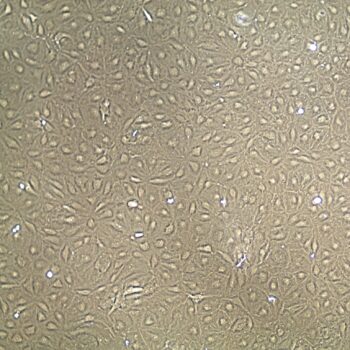
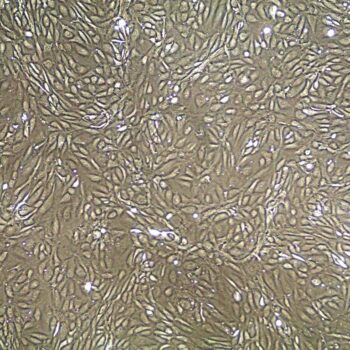
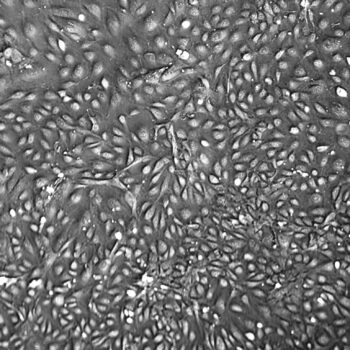
Normal Human Primary Endothelial Cells
Primary cells are directly cultured from their source organ tissue, for true experimental outcomes. Choose cells representative of the general population or select by donor demographics.
Human Primary Endothelial Cells include: aortic, coronary artery, pulmonary artery, iliac artery, HUVEC (human umbilical vein), dermal microvascular, cardiac microvascular, and lung microvascular.
Lifeline® normal Human Endothelial Cells provide an ideal cell system to study angiogenesis, cancer, wound healing, atherosclerosis, inflammation or vascular biology. Additionally, these cell lines can also be used for drug development or screening.
Lifeline® Human Endothelial Cells are not exposed to antimicrobials or phenol red when cultured in Lifeline® VascuLife® low-serum medium, to avoid cell stress and “masking effects” that may negatively impact experimental results.
Primary Endothelial Cells can be cultured in VascuLife® low-serum medium, with or without human VEGF. This medium has been optimized specifically for primary endothelial cells to maximize cell life expectancy.