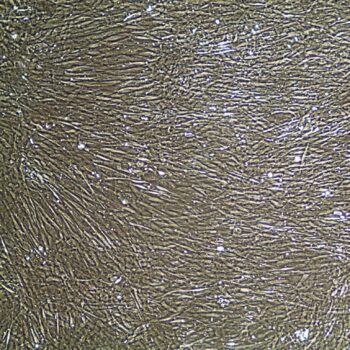
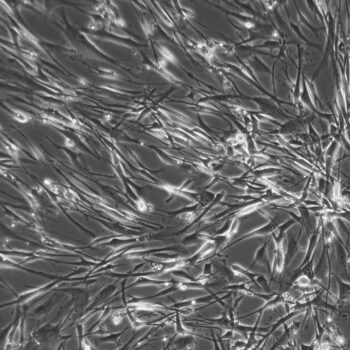
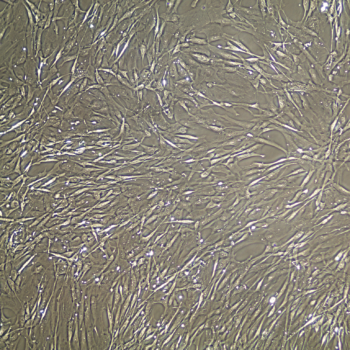
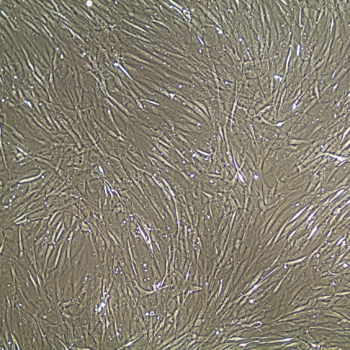
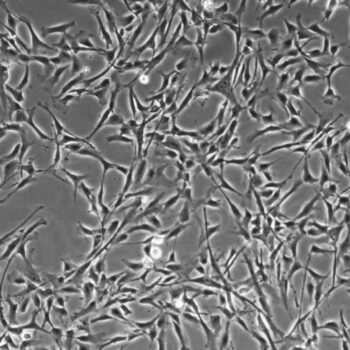
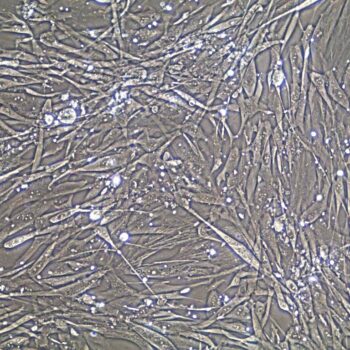
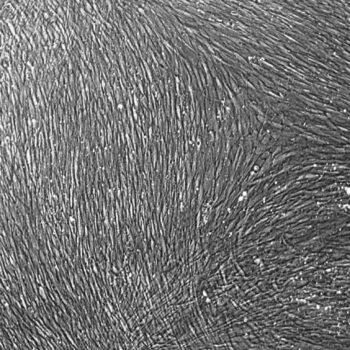
Primary Human Smooth Muscle Cells, Normal
Normal human primary smooth muscle cells include: airway, aortic, bladder, coronary artery, prostate, pulmonary artery, and uterine.
- Primary smooth muscle cells are cells that are directly cultured from their source organ tissue.
Lifeline® Primary Human Smooth Muscle Cells (HSMC), when grown in Lifeline VascuLife SMC Medium, provide an ideal low serum (5%) culture model for the study of angiogenesis, atherosclerosis, diabetes or vascular/pulmonary biology.
Primary smooth muscle cells are cryopreserved to ensure the highest viability, purity and plating efficiency. Our HSMC are quality tested in VascuLife SMC Medium and demonstrate optimal low serum growth over a period of at least 15 population doublings at rates equal to or greater than other serum-supplemented media.
Lifeline primary smooth muscle cells are not exposed to antimicrobials or phenol red when cultured in VascuLife SMC Medium, an advantage since these supplements can cause cell stress and “masking effects” that may negatively impact experimental results.