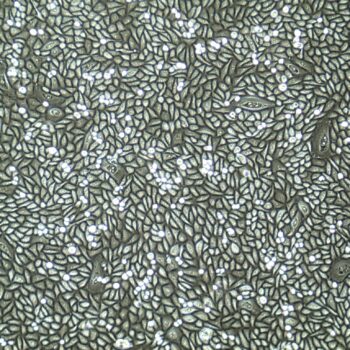
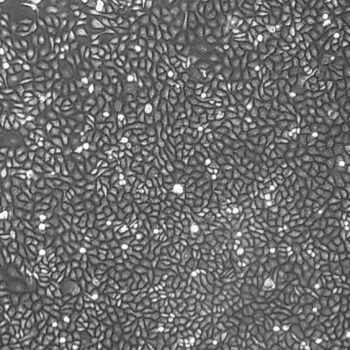
Human Oral Keratinocytes (Gingiva)
$917.30
In stock
Product Description
Lifeline® Oral Keratinocytes (Gingiva) are normal human Primary Cells. When grown in Lifeline® DermaLife K Keratinocytes Medium, Oral Keratinocytes provide an ideal serum-free culture model for the study of microbial-to-epithelial cell interactions, wound healing, toxicology or oral epithelial biology.
These Primary Keratinocytes come from a wide variety of donors, without the accumulated mutations found in immortal cell lines. Primary Keratinocytes are Directly Cultured from their source organ tissue without any modifications. So you can be assured of the best possible, valid research outcomes.
Primary Oral Keratinocytes have been isolated, plated and expanded in culture vessels twice before being harvested for cryopreservation to ensure the highest viability and plating efficiency.
Human oral keratinocytes are quality tested in DermaLife K Medium to ensure proper growth and morphology over a period of at least 15 population doublings. DermaLife K is an optimized medium, composed and supplemented to maximize the life expectancy of Primary Oral Keratinocytes.
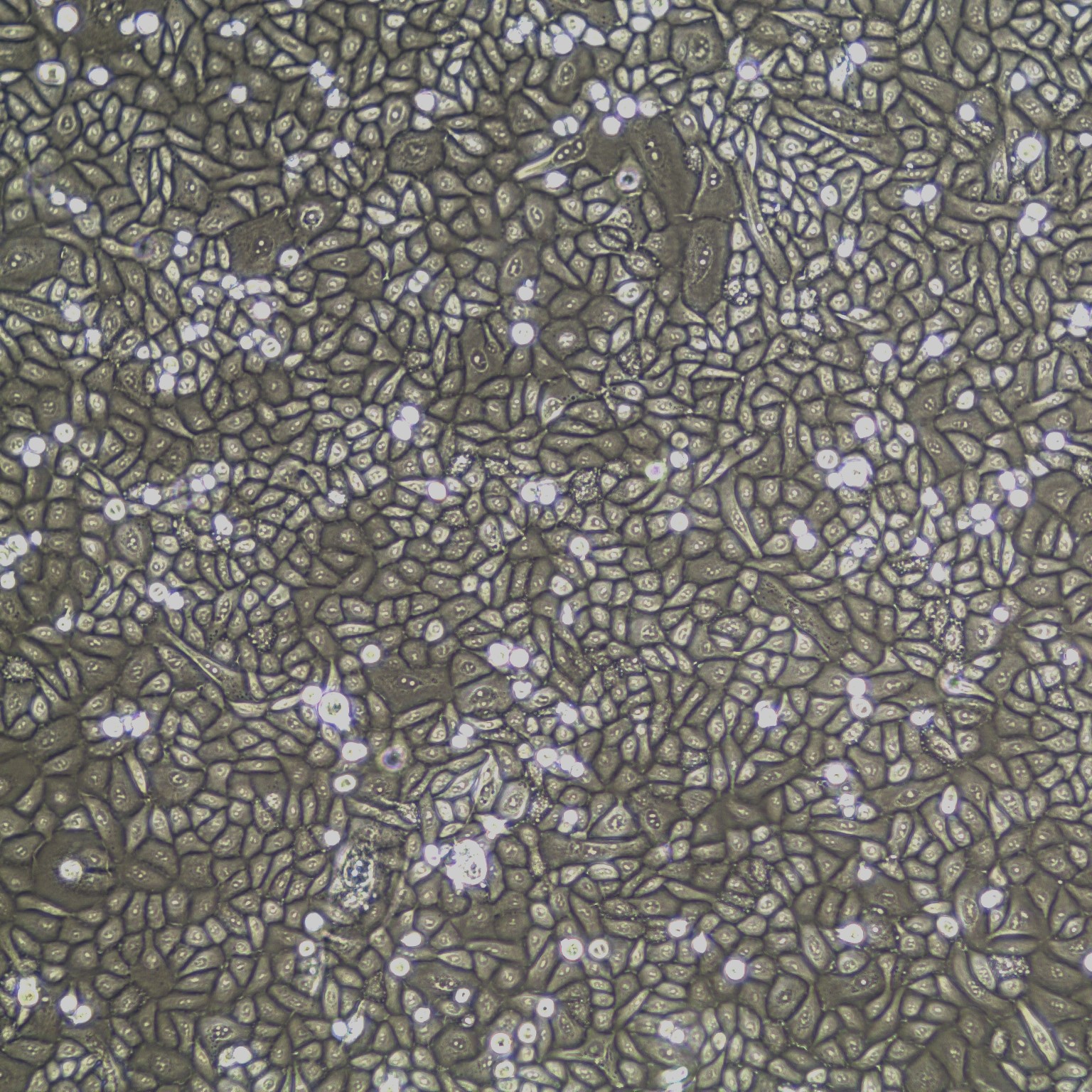
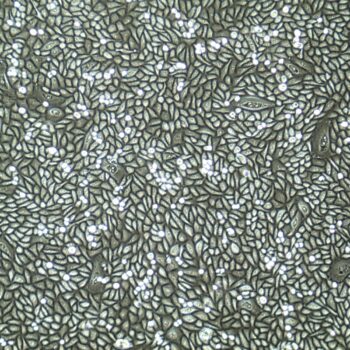
Human Oral Keratinocytes Gingiva), passage 3,
5 days after inoculation with 2,500 cells/cm2 (100X)
Lifeline keratinocytes are not exposed to antimicrobials or phenol red when cultured in the respective Lifeline medium. This is an advantage since these supplements can cause cell stress and “masking effects” that may negatively impact experimental results.
Normal human oral keratinocytes (gingiva) are tested for:
- Cell count: 500,000 cryopreserved cells per vial
- Proliferation and Morphology: Normal cell appearance for 15 population doublings
- Cell viability: Minimum 70% viability when thawed from cryopreservation
- Sterility testing: Negative for mycoplasma; negative for bacterial and fungal growth for 14 days
- Virus testing: Negative for HIV-1, HIV-2, HBV, and HCV by PCR
Product Specification Sheets
Keratinocyte Systems
Complimentary Products
- DermaLife K Keratinocyte Calcium-Free Medium Kit [LL-0029]
- DermaLife K Keratinocyte Medium Complete Kit [LL-0007]
- Human Gingival Fibroblasts [FC-0095]
SDS and “Safety Data Sheets”
(MSDS or Material Safety Data Sheets)
Cells
Product Specification Sheets
Oral Keratinocytes - Human