VascuLife® EnGS-Mv Microvascular Endothelial Kit
$181.90
In stock
Product Description
VascuLife® EnGS-Mv Medium (containing Endothelial Cell Growth Supplement) is optimized for the culture of Human Primary Microvascular Endothelial Cells.
- VascuLife culture medium supports the rapid proliferation of these Primary Microvascular Endothelial Cells in a 5% serum environment.
Primary Microvascular Cells cultured in VascuLife medium out-perform Primary Cells grown in any similar medium.
You’ll get consistently reproducible results with VascuLife Microvascular Endothelial medium, because it’s balanced and supplemented expressly for Primary Microvascular Endothelial Cells to maximize that individual Primary Cell type’s life expectancy.
- Your Primary Microvascular Endothelial Cells will plate better using VascuLife optimized media (as compared to our nearest competitors’ media or traditional high-serum media)
- Your Primary Cells will grow more quickly in VascuLife medium than in other commercially-available media
- Primary Microvascular Endothelial Cells will maintain better morphology when cultured in VascuLife media
VascuLife Basal Medium contains no antimicrobials and no phenol red.
VascuLife EnGS-Mv is offered in a kit format, composed of basal medium (in a light-protected 500 mL bottle). This kit allows you to prepare fresh complete medium in your laboratory, extending shelf life and enhancing performance.
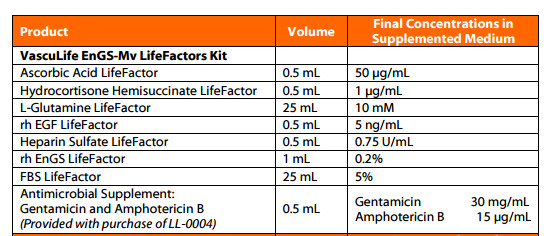
The LifeFactors Kit with associated supplements and growth factors.includes:
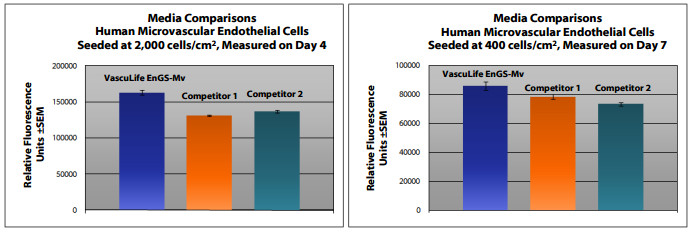
VascuLife EnGS-Mv Medium grows Human Primary Microvascular Endothelial Cells, without the addition of human VEGF, at rates that meet or exceed other commercially available serum-containing media while maintaining excellent cell morphology. In comparisons with other commercially-available media, VascuLife shows superior proliferation at different seeding densities.
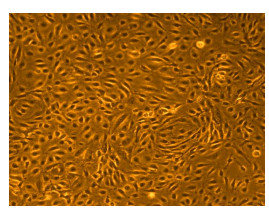
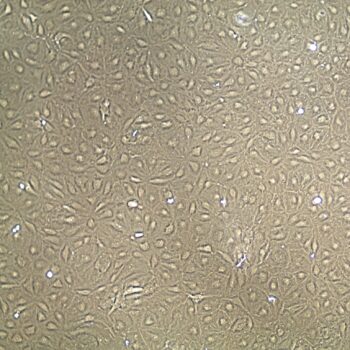
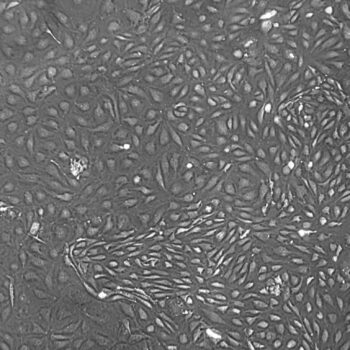
Shown above: Human Primary Dermal Microvascular Endothelial Cells, passage 2, 4 days after inoculation with 5,000 cells/cm2 (100X).
Shown below: Human Primary Dermal Microvascular Endothelial Cells inoculated at low seeding density in 24-well plates. Higher fluorescence represents a greater number of cells per well, and therefore, better proliferation.
Extensive Testing for Guaranteed Consistency and Reproducible Results
VascuLife EnGS-Mv culture medium is quality tested for:
- Sterility: Negative for bacteria and fungal growth
- pH: 7.8 +/- 0.3
- Osmolality: 270 +/- 10 mOsm
- Endotoxin: < 0.5 EU/mL
Our ISO-style quality assurance ensures lot-to-lot consistency in every production run.
Product Specification Sheets
Endothelial Cell Systems
Skin Cell Systems
Complimentary Products
Product Specification Sheets
Endothelial Cells - Human
Endothelial Cell Systems
VascuLife™
Kit Components
- LM-0002
- LS-1035