Normal Human Bone Marrow Mesenchymal Stem Cells
$1,002.30
In stock
Product Description
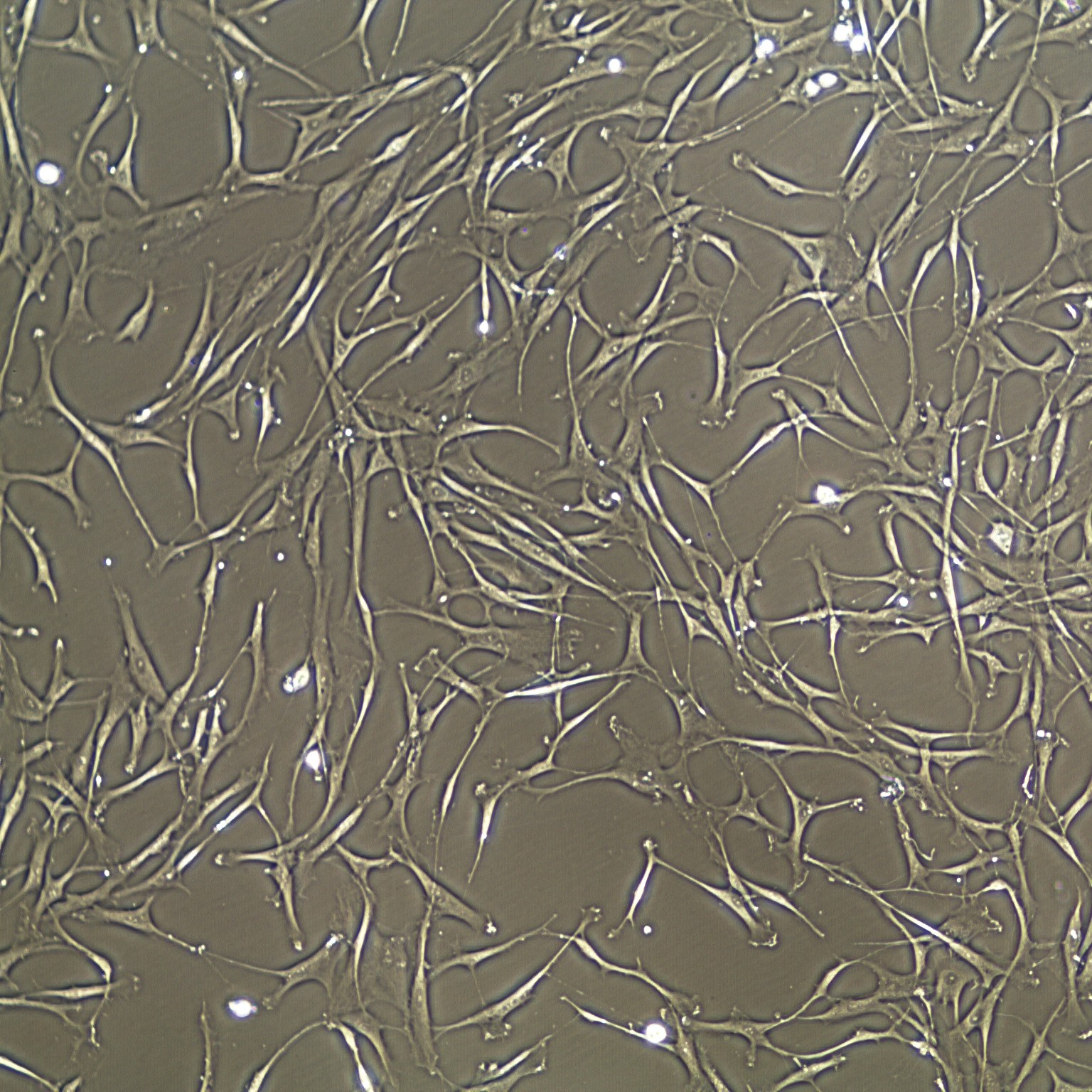
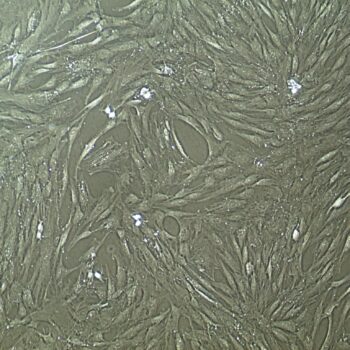
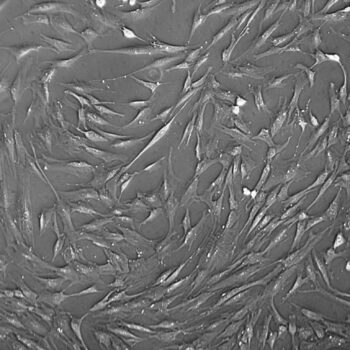
Lifeline® Human Bone Marrow Mesenchymal Stem Cells (HMSC-BM) provide an ideal culture model for the study of multipotent stem cell biology and differentiation processes.
HMSC-BM are isolated from Bone Marrow aspirate, expanded once, and cryopreserved as secondary cells to ensure optimal phenotype and the highest viability and plating efficiency.
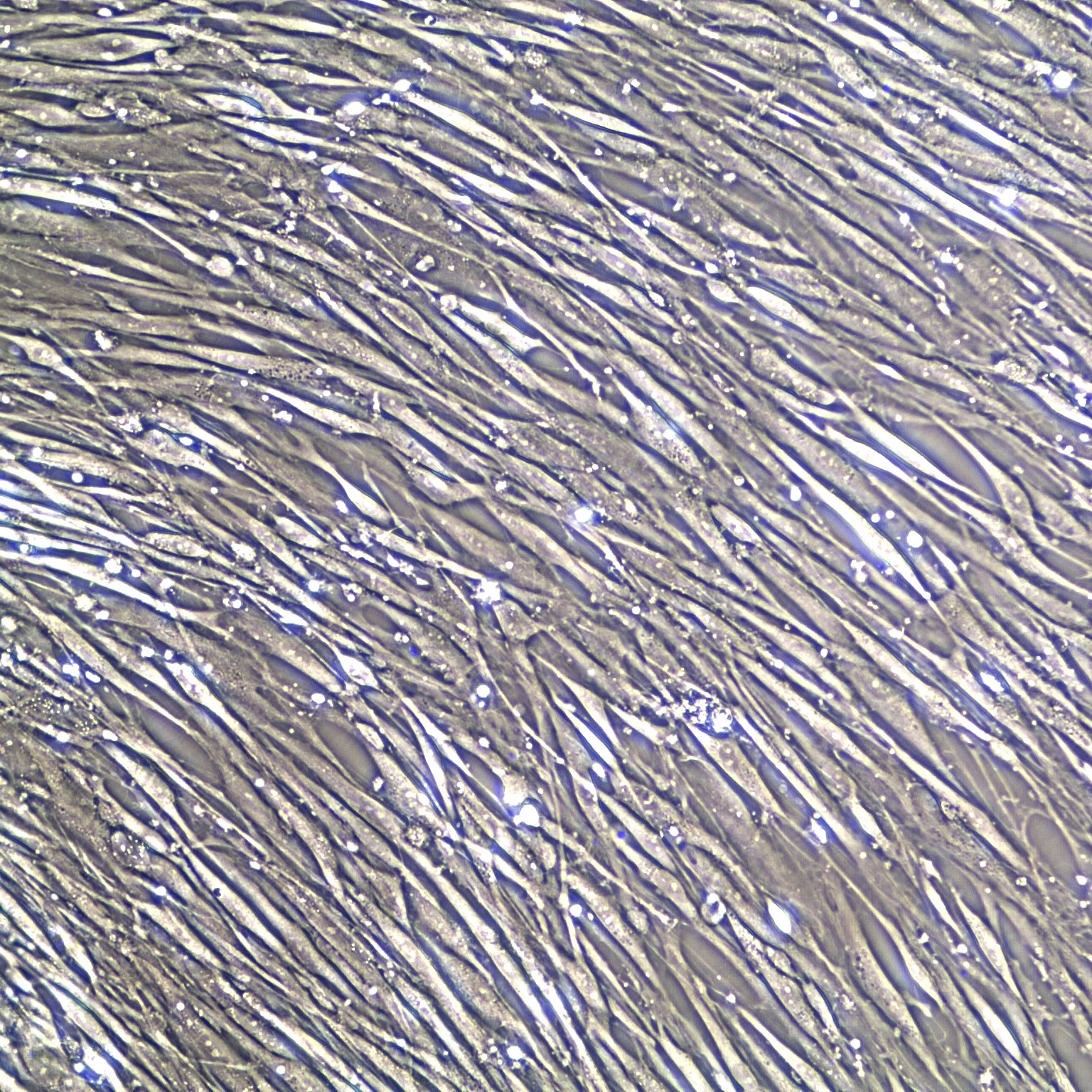
Bone Marrow Mesenchymal Stem Cells can be expanded in an undifferentiated state for future differentiation to multiple lineages.
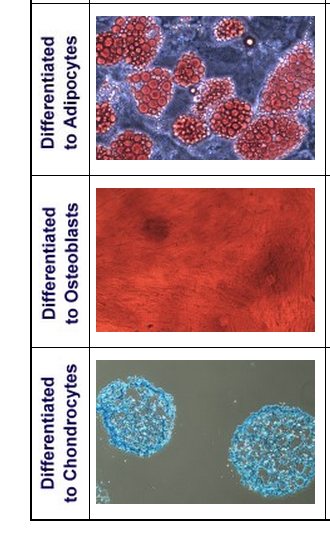
- Lifeline Human Mesenchymal Stem Cells — Bone Marrow may be differentiated down the typical mesenchymal lineages, such as adipogenic, chondrogenic, and osteogenic.
Human Mesenchymal Stem Cells-Bone Marrow are characterized by flow cytometry to ensure the proper expression of multiple markers of mesenchymal stem cells. They are uniformly positive for CD29, CD44, CD73, CD90, CD105, and CD166. They are uniformly negative for CD14, CD31, CD34, and CD45.
Lifeline has developed optimized Mesenchymal Stem Cell Bone Marrow Culture Media to expand Bone Marrow Mesenchymal Stem Cell products in the undifferentiated state, as well as optimized media kits for inducing Adipogenesis, Chondrogenesis, and Osteogenesis. Additionally, Lifeline provides convenient staining kits for staining the fully differentiated cells.
Mesenchymal Stem Cells -Bone Marrow are quality tested for:
- Morphology: Normal morphology for 3 passages
- Cell Viability: Minimum 70% viability when thawed from cryopreservation
- Sterility Testing: Negative for mycoplasma. Negative for bacterial and fungal growth
- Virus Testing: Negative for HIV-1, HIV-2, HBV, and HCV by PCR
- Specific Staining: Positive* for CD29, CD44, CD73, CD90, CD105, CD166.
Negative* for CD14, CD31, CD34, CD45.
*Lifeline defines positive expression as when greater than 95% of the cell population expresses that cell marker.
*Lifeline defines negative expression as when less than 2% of the cell population expresses that cell marker.
Product Specification Sheets
Stem Cell Systems
Staining Kits
Complimentary Products
SDS and “Safety Data Sheets”
(MSDS or Material Safety Data Sheets)
Cells
Product Specification Sheets
Mesenchymal Stem Cells - Human
ChondroLife™
AdipoLife™
OsteoLife™