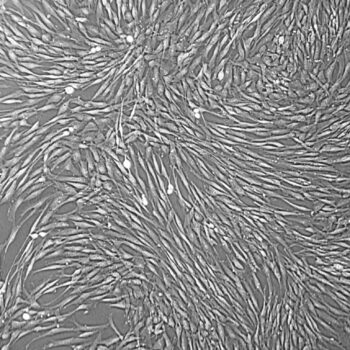
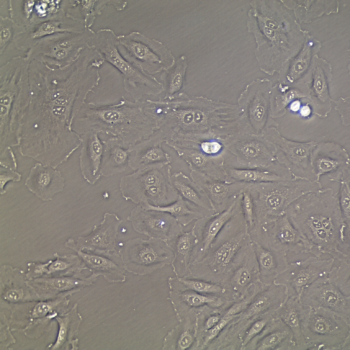
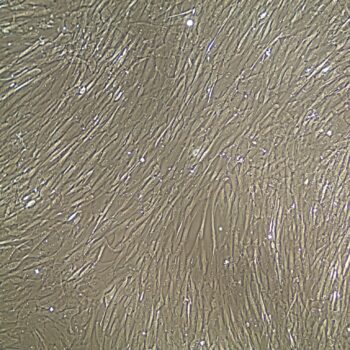
Human Umbilical Vein Endothelial Cells (HUVEC) –10-Donor Pool
$283.80
In stock
Product Description
Lifeline® Primary Normal HUVEC 10-Donor Pools, when grown in Lifeline VascuLife® Endothelial Culture Medium, provide an ideal low
-serum culture model, with or without human VEGF, for the study of angiogenesis, atherosclerosis or vascular biology without the concern of atypical results from a single donor.
- Primary Human Umbilical Vein Endothelial Cells can be grown in low serum (2%) medium without phenol red or antimicrobials when cultured in VascuLife medium.
- Our Primary HUVEC may be cultured with or without VEGF.
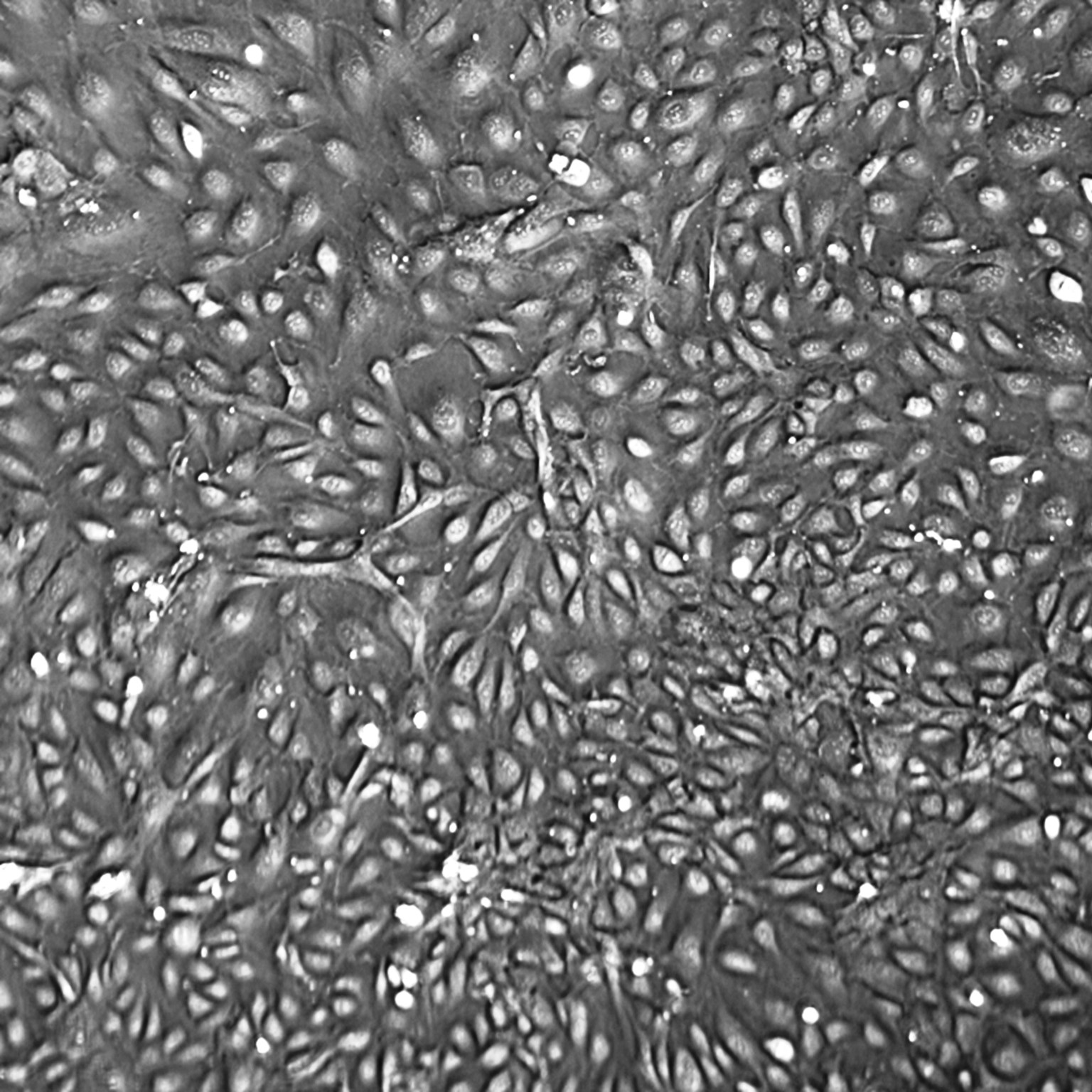
Lifeline Primary Human Umbilical Vein Endothelial Cells are isolated from human umbilical cords. The cells are plated and expanded in culture vessels twice before being harvested for cryopreservation to ensure the highest viability, purity and plating efficiency.
- They are isolated in VascuLife EnGS Medium to ensure that they remain unexposed to recombinant human VEGF.
Our HUVEC are quality tested in VascuLife EnGS Medium and demonstrate optimal reduced-serum growth over a period of at least 15 population doublings at rates equal to or greater than other serum-supplemented media.
Lifeline Human Umbilical Vein Endothelial Cells are not exposed to antimicrobials or phenol red when cultured in VascuLife medium, an advantage since these supplements can cause cell stress and “masking effects” that may negatively impact experimental results.
500,000 cells per vial
Lifeline Primary HUVEC are quality tested for:
- Proliferation and Morphology: Normal cell appearance for 15 population doublings
- Cell Viability: Minimum 70% viability when thawed from cryopreservation
- Sterility: Negative for mycoplasma. Negative for bacterial and fungal growth
- Virus: Negative for HIV-1, HIV-2, HBV, and HCV by PCR
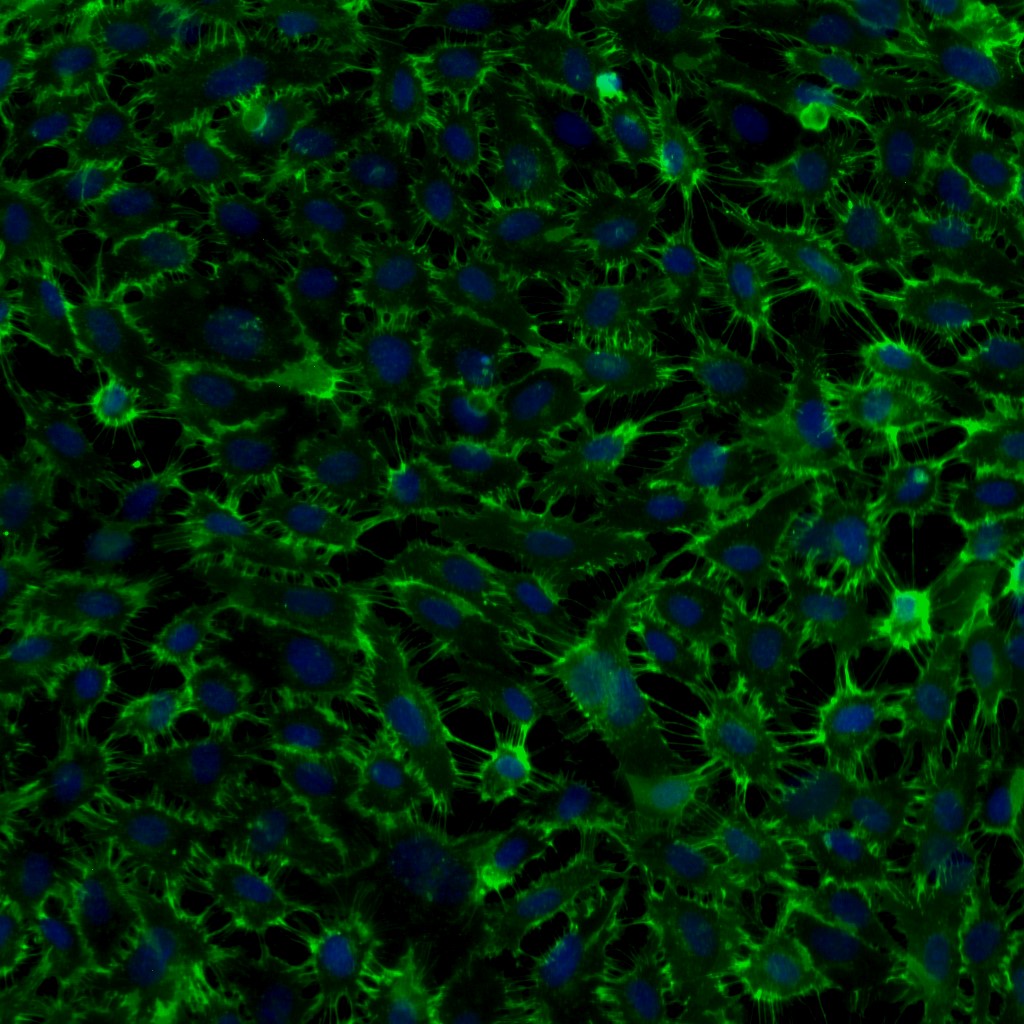
- Specific Staining: von Willebrand Factor positive. Smooth muscle a-actin negative.
Product Specification Sheets
Endothelial Cell Systems
Complimentary Products
SDS and “Safety Data Sheets”
(MSDS or Material Safety Data Sheets)
Cells
Product Specification Sheets
Endothelial Cell Systems